Hyperlipidemia: Etiology, Pathophysiology, Symptoms, Diagnosis, Treatment, Prognosis
Hyperlipidemia is an umbrella term that refers to any of several acquired or genetic disorders that result in a high level of lipids (fats, cholesterol and triglycerides) circulating in the blood.
Introduction
Hyperlipidemia is a condition that incorporates various genetic and acquired disorders that describe elevated lipid levels within the human body. Hyperlipidemia is extremely common, especially in the Western hemisphere, but also throughout the world. Alternatively, a more objective definition describes hyperlipidemia as low-density lipoprotein (LDL), total cholesterol, triglyceride levels, or lipoprotein levels greater than the 90th percentile in comparison to the general population, or an HDL level less than the 10th percentile when compared to the general population. Lipids typically include cholesterol levels, lipoproteins, chylomicrons, VLDL, LDL, apolipoproteins, and HDL.
Through a vast array of trials and studies, it has been consistently shown that elevated levels of LDL cholesterol increase a person’s risk for the development of atherosclerotic plaques and subsequent vascular disease. In stark contrast, high-density lipoprotein (HDL) cholesterol assists in regulating cholesterol levels to prevent imbalances that would increase the risk of atherosclerotic vascular disease.
Each patient’s LDL cholesterol goal is conditional on their overall cardiovascular risk, and medical therapy should be independently tailored to the patient. Managing risk factors, such as hyperlipidemia, to diminish the risk for atherosclerotic cardiovascular disease is referred to as “primary prevention.”
The grounds for lowering LDL cholesterol derives from widespread epidemiologic data that reveals a positive, continuous correlation between LDL cholesterol levels, cardiovascular events, and patient mortality.
Treatment of hyperlipidemia continues to evolve as we better conceptualize the underlying pathophysiology, and we concurrently improve on preceding medical therapies. This article will overview the background, diagnosis, and most recent treatment guidelines for hyperlipidemia.
Etiology
Hyperlipidemia subdivides into two broad classifications: primary (familial) or secondary (acquired) hyperlipidemia.
Primary hyperlipidemia derives from a plethora of genetic disorders that a patient may inherit through birth, while secondary hyperlipidemia typically originates from an alternate underlying etiology, such as an unhealthy diet, medications (amiodarone, glucocorticoids), hypothyroidism, uncontrolled diabetes, and/or a poor lifestyle regimen.
Underlying disturbances in lipoprotein metabolism are often familial, making a patient’s family history that much more valuable. For example, about 54 percent of patients (in one study) with a history of premature coronary artery disease had an underlying hereditary disorder.
In most patients, hyperlipidemia has a polygenic inheritance pattern, and manifestations of the disorder are largely influenced by secondary factors such as (central) obesity, saturated fat intake, and the cholesterol content within a person’s diet. Various less conventional risk factors will also appear below.
Cholesterol is the circulating fatty substance, most implicated in the atherogenic process. Its origin is twofold: 300 to 700 mg per day is of exogenous origin, that is, coming from an excessive intake of dietary fats, especially of animal origin; 800 to 1200 mg per day is the work of an endogenous synthesis, in particular the liver.
In addition to excessive consumption of animal fats, other frequent causes of hypercholesterolemia and/or increase in triglycerides are diabetes, chronic renal failure, nephrotic syndrome, hypothyroidism, age, sedentary lifestyle.
Other iatrogenic causes may be the intake of certain drugs such as thiazide diuretics, beta-blockers, estrogen-progestin contraceptives, antiretrovirals.
Genetic dyslipidaemias, much rarer, are the basis of the changes in the blood lipid rate in the measure of about 60% and are often responsible for cardiovascular diseases at an early age.
Epidemiology
There are over three million adults throughout the United States and Europe that currently have a diagnosis of hyperlipidemia, and that number continues to rise at a drastic pace. Hyperlipidemia is typically a chronic, progressive disease process that demands lifestyle and dietary changes, with the potential need for additional lipid-lowering medications.
The degree of hyperlipidemia is highest in patients with premature coronary artery disease (CAD), defined as CAD arising in males before age 55 to 60 years and females before age 65 years. Under the prior specified circumstances, the incidence of hyperlipidemia is around 75-85%, opposed to roughly 40 to 48% in the control population of comparable age, but without the presence of premature coronary artery disease.
Estimates are that over 50% of American adults have elevated LDL levels, and it is speculated that under 35% of those patients adequately manage their high LDL levels, clearly depicting an undertreated disease.
Per the JAMA Network, “Prevalence of dyslipidemia was significantly greater among whites than blacks (women, 64.7% vs. 49.5%; and men, 78.4% vs. 56.7%; P<.001 for both) and amongst men than women (P≤.02 in every ethnic group).”
Intuitively, in countries with lower overall rates of obesity and saturated fat consumption, the prevalence of hyperlipidemia and subsequent coronary artery disease is lower, when contrasted to rates in Europe and throughout the United States.
Children under two years of age, if underweight or obese, may develop secondary (non-genetic) pediatric hyperlipidemia.
Pathophysiology
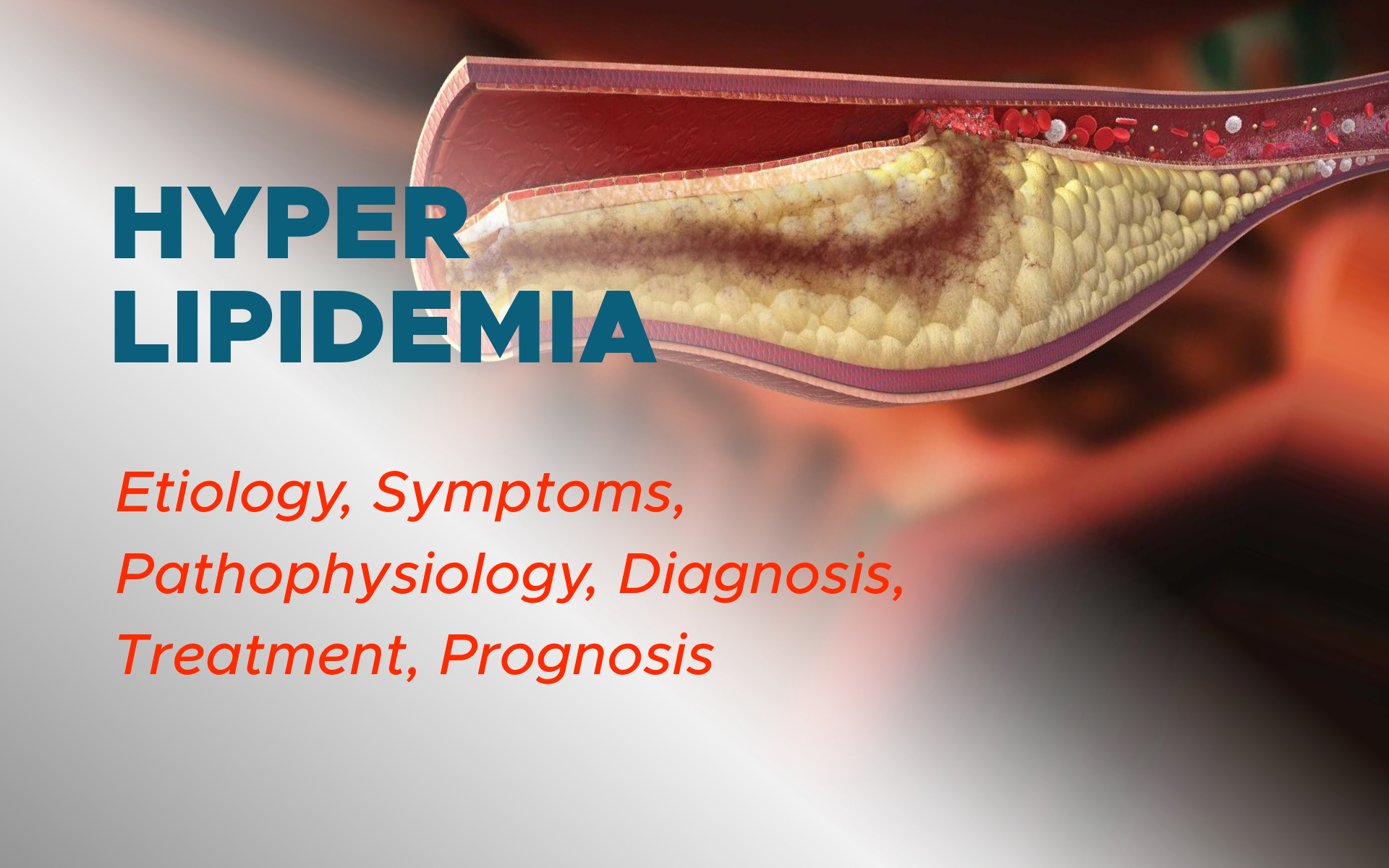
Hyperlipidemia, in particular elevated LDL (hypercholesterolemia), is one of the most prevalent risk factors contributing to the evolution of atherosclerosis and consequent vascular disease. It is simply defined as elevated concentrations of lipids or fats within the blood.
Numerous factors contribute to the development of atherosclerosis, including endothelial damage, hyperlipidemia, inflammatory and immunologic factors, plaque erosion or rupture, hypertension, and smoking.
Atherosclerosis frequently remains asymptomatic until plaque stenosis reaches 70 to 80% of the vessel’s diameter. Atherosclerosis originates after underlying endothelial damage occurs, which appears to stem from the loss of nitric oxide within the endothelium. This process leads to increased inflammation directly around the site of dysfunction, permitting the accumulation of lipids within the innermost layer of the endothelial wall.
The lipids are then engulfed by macrophages, leading to the establishment of “foam cells.” This cholesterol build-up within the “foam cells” causes subsequent mitochondrial dysfunction, apoptosis, and, ultimately, necrosis of the underlying tissues.
Smooth muscle cells encapsulate the pack of “foam cells” or debris, which produces a fibrotic plaque that inhibits the underlying lipids (debris) from being destroyed.
Tissue factor, alongside increased platelet activity, is known as a primary initiator of coagulation, which increases the risk for plaque rupture and thrombosis.

Atherosclerotic plaques evolve via two distinct mechanisms: a slower, chronic plaque build-up that progressively leads to luminal stenosis, versus an acute onset of rapid luminal obstruction secondary to plaque rupture and thrombosis. Both mechanisms are capable of causing clinically significant disease that should be addressed by a physician as soon as possible.
For most patients, hyperlipidemia is polygenic in inheritance, and the manifestation of the disorder is considerably influenced by factors such as (central) obesity, saturated fat intake, and the cholesterol content within a person’s diet.
Another mechanism involves elevated levels of “apo B-100” lipoproteins within the plasma, which may lead to atherosclerotic disease, even when the patient has no other risk factors. It is often that there is a combination of genetic and environmental factors at play that ultimately contribute to a person’s risk of developing hyperlipidemia and cardiovascular disease.
Many systemic diseases, which stimulate an inflammatory sub-layer with clinical or sub-clinical values, can cause dyslipidemia and atherosclerotic problems. Some examples:
-
Psoriasis
-
Crohn disease
-
Inflammatory bowel disease
-
Chronic obstructive pulmonary disease
-
Depression
-
Chronic pain
-
Pediatric alopecia areata
-
Chronic kidney disease
Histopathology
In the presence of hyperlipidemia, not only the vascular structures are negatively involved but also other tissues. Fort example, research has demonstrated through patellar tendon shear wave velocities that there is a direct relationship between the intrinsic alteration of the patellar tendon and the presence of hyperlipidemia.
The tendon becomes stiffer with morphological alteration of the tissue and changes in the type of cells present. Increases the number of macrophages in the tendon tissue, damaged collagen fibers, replacement of collagen cells with lipid cells; collagen type III increases, which is less elastic, matrix metalloproteinases increase. The tendon in the presence of hyperlipidemia becomes mechanically less effective and more prone to injury.
History and Physical
Regularly, patients presenting with underlying hyperlipidemia remain asymptomatic, therefore obtaining a precise and thorough history is essential.
Upon taking a patients history, it is crucial to obtain a profound understanding of each patient’s family history of cardiovascular disease, hyperlipidemia, and/or familial hypercholesterolemia; their diet and exercise habits; tobacco, alcohol, or drug use; the presence of coronary artery disease; risk factors or history of CAD; and/or symptoms of peripheral arterial disease or angina.
In addition to obtaining a detailed history, a focused physical exam is also very important. Obtaining accurate blood pressure measurements, observing the patients skin for xanthomas, listening for carotid and femoral bruits for evidence of stenosis, listening for an S4 heart sound, and palpating for intact peripheral pulses in all four extremities are fast and simple physical exam findings that can assist in your diagnosis of hyperlipidemia.
Evaluation
Various experts have developed lipid screening guidelines that include the “lipid profile” to measure cholesterol and triglyceride levels. Guidelines differ as to what age primary providers should start screening and how often they should screen patients for hyperlipidemia.
In general, it is advised that routine lipid screening should occur when a male turns 35 years of age (if no other cardiovascular risk factors) or 25 years of age (if the patient has other cardiovascular risk factors). It is also suggested that routine lipid screening be initiated in females at 45 years of age (if no other cardiovascular risk factors present) or 30 to 35 years of age (if the patient has other cardiovascular risk factors).
For lower-risk patients, lipid screening every five years is reasonable, and more frequent screening is recommended as the patient’s cardiovascular risk increases.
As stated above, the most valuable laboratory test to collect is checking fasting lipid profile, which routinely includes LDL, HDL, triglycerides, and total cholesterol. A v-LDL, total cholesterol: HDL, and LDL: HDL ratios can be added on for a more comprehensive test. It is necessary to not eat or drink anything besides water for 9 to 12 hours as not to skew the results of the lipid panel (mainly the triglyceride levels).
Before starting a statin for high LDL levels, it is important to obtain liver function tests to ensure there is no prior liver dysfunction, as statins may exacerbate this issue. For risk stratification purposes, a Hgb A1c level is necessary to screen for diabetes mellitus, and the clinician should always examine blood pressure measurements to ensure the patient does not have underlying hypertension.
Additionally, a TSH should be ordered to rule out underlying thyroid abnormalities, and a simple urinalysis can be collected to screen for albuminuria. These tests are critical for risk-stratification of your patient to accurately assess the potential risks versus benefits of initiating medical therapy in a patient with hyperlipidemia.
Treatment / Management
The decision to treat elevated LDL cholesterol levels depends on the determination of overall cardiovascular risk by the patient’s primary physician, and this should be discussed in great detail with the patient. The absolute risk reduction affiliated with lipid-lowering therapy for hyperlipidemia is generally less than for patients with known underlying cardiovascular disease.
To reduce risk in patients without a known diagnosis of cardiovascular disease, only treatments of elevated LDL cholesterol have proven to be of clinical benefit. There is no proven clinical benefit to the treatment of hypertriglyceridemia or low HDL cholesterol levels.
Initial treatment modalities are focused on diet and lifestyle modification, with the possible addition of lipid-lowering medications if needed. Patients with mild hyperlipidemia and low ASCVD risk (below 7.5% 10-year risk) should focus on a low fat, low carbohydrate diet, and moderate to high-intensity physical activity (recommended 30 minutes per day, 5 to 6 days per week).
The AHA advises limiting saturated fat consumption to about 5% of your daily calories and restricting the total quantity of trans-saturated fat consumption as much as possible. Quitting smoking, lowering blood pressure, and losing weight have all proven to be very advantageous in regards to lowering vascular disease risk.
For patients at moderate to high ASCVD risk (over 7.5% 10-year risk), the addition of lipid-lowering “statin” medications should be added.
The most well rounded and complete meta-analysis investigating primary prevention trials in hyperlipidemia patients discovered an all-cause mortality benefit and that lowering LDL cholesterol is effective at decreasing cardiovascular events, in particular, reducing the risk of myocardial infarction.
There is a clear and proven benefit to statin therapy for the vast majority of patients, from low risk to high risk, and if side effects and financial constraints did not exist, almost all patients would be prescribed statin therapy. Therefore, these medication’s side effects and costs should be weighed against the individual patient’s potential benefit from taking the drug.

Key Recommendations for Practice: SORT evidence rating system:
- Patients with a high risk of ASCVD (>7.5% 10-year risk) should receive statin therapy for primary prevention: Rating B.
-
Statin therapy should be initiated for secondary prevention in patients with known ASCVD, absent any contraindication: Rating A
-
Niacin, fibrates, and omega-3 fatty acids should not be routinely given for primary or secondary prevention of ASCVD: Rating A
-
A moderate-intensity statin plus ezetimibe should merit consideration as an alternative in patients with acute coronary syndrome who cannot tolerate high-intensity statin therapy: Rating B.
-
Moderate-intensity statins include: lovastatin 40 mg, pravastatin 40 mg, simvastatin 40 mg, atorvastatin 10 to 20 mg, and rosuvastatin 5 to 10 mg
-
High-intensity statins include: atorvastatin 40 to 80 mg, rosuvastatin 20 to 40 mg
If the provider and patient reach a mutual decision to initiate medical therapy with statins, the overall risk reduction in cardiovascular events is customarily around 20 to 30% in most clinical trials.
The commonly referred to trials of which the majority of guidelines are based, included pravastatin 40 mg, lovastatin 20 to 40 mg, atorvastatin 10 mg, and rosuvastatin 10 mg, so general recommendations are to choose one of these statins listed.
It is always important to schedule close follow up with patients that are starting lipid-lowering statin therapy. The vast majority of benefits arising from statin therapy originates from the moderate dose, with a much more diminutive benefit deriving from the addition of high-intensity therapy.
However, the benefit of high-intensity therapy remains clinically significant and should merit consideration for all high-risk patients.
If a patient encounters an allergy or intolerance to a statin medication, the suggestion is to reduce the original or to transition to a different lipid-lowering medication altogether.
Among patients that have reported muscle-related statin intolerances, according to one 24 week trial, evolocumab (PCSK9 inhibitor) reduced LDL cholesterol levels significantly more when compared to ezetimibe. However, both of these medications are reasonable secondary options for treatment.
Differential Diagnosis
It is imperative for diagnosing providers to assemble a comprehensive list of differentials when screening a patient for hyperlipidemia. Primary disorders should always be considerations; these include familial hypercholesterolemia, familial combined hyperlipidemia, dysbetalipoproteinemia, familial defective apo B-100, and PCSK9 gain of function mutations.
Then, these secondary disease processes must also be in the differential when diagnosing a patient with hyperlipidemia: obstructive liver disease or biliary obstruction, hypothyroidism, nephrotic syndrome, chronic renal insufficiency, anorexia, obesity, metabolic syndrome, and diabetes.
A full history and physical complete with comprehensive labs should be obtained to narrow down the differential and make the correct diagnosis.
Prognosis
Hyperlipidemia is often a life-long disease process, but one that is typically quite manageable. However, if hyperlipidemia is left untreated, the disease is progressive and will often lead to severe underlying vascular disease processes, which can prove fatal.
Ongoing persistent exposure to high serum lipid levels throughout early adulthood increases the person’s subsequent risk of coronary heart disease in a dose-dependent fashion.
Adults with ongoing exposure to moderate or severe elevations in non-HDL cholesterol levels have concurrent elevated risks for developing coronary heart disease and would likely benefit from aggressive medical treatment modalities, including, which includes high-intensity statin therapy in addition to diet and lifestyle modifications.
Based on a 20 year follow up of the “West of Scotland Coronary Prevention Study,” the researchers noted that patients who received statin therapy for five years had improved survival rates and a clinically significant reduction in cardiovascular disease over the 20 years.
This data supports the numerous other studies and trials that also reveal a significant cardiovascular risk reduction when using statin therapy appropriately, and there was the implementation of a proactive treatment approach.
Complications
Complications from undertreated or untreated hyperlipidemia include all types of vascular disease, which may prove fatal down the road. These include, but are not limited to, coronary artery disease, peripheral artery disease, cerebrovascular accidents, aneurysms, type II diabetes, high blood pressure, and even death.
Statin medication complications include myopathy, renal injury, arthralgia, extremity pains, nausea, myalgia, elevated liver enzymes/hepatotoxicity, diarrhea, and rhabdomyolysis. Reportedly up to 5 to 20% of patients taking a statin medication experience a muscle-related intolerance.
A lower dose of the statin should be attempted and/or attempting to transition to another lipid-lowering medication such as ezetimibe or evolocumab.
Postoperative and Rehabilitation Care
Aerobic exercise has shown to increase the levels of the HDL-cholesterol protein, with an anti-atherogenic action: the HDL that transports cholesterol from the walls of the arteries and peripheral tissues to the liver and the same HDL has an antioxidant and anti-inflammatory capacity that induces the release of nitric oxide (vasodilator compound).
Estimates are that adding 10 minutes of physical activity to daily exercise increases the concentration of HDL by 1.4 mg/dl, and it has been calculated that, on average, an adequate training program can increase cholesterol HDL of 4.6%.
A combination of aerobic and anaerobic activity is equally capable of positively influencing blood lipid levels.
One hour per week of resistance training (anaerobic activity) can improve the lipid profile and corresponds to a correct indication in order not to exceed in physical effort and maintain optimal health.
References
- Fredrickson DS. An international classification of hyperlipidemias and hyperlipoproteinemias. Ann Intern Med. 1971 Sep;75(3):471-2.
- Ballantyne CM, Grundy SM, Oberman A, Kreisberg RA, Havel RJ, Frost PH, Haffner SM. Hyperlipidemia: diagnostic and therapeutic perspectives. J Clin Endocrinol Metab. 2000 Jun;85(6):2089-112.
- Faxon DP, Fuster V, Libby P, Beckman JA, Hiatt WR, Thompson RW, Topper JN, Annex BH, Rundback JH, Fabunmi RP, Robertson RM, Loscalzo J., American Heart Association. Atherosclerotic Vascular Disease Conference: Writing Group III: pathophysiology. Circulation. 2004 Jun 01;109(21):2617-25.
- Vodnala D, Rubenfire M, Brook RD. Secondary causes of dyslipidemia. Am J Cardiol. 2012 Sep 15;110(6):823-5.
- Stamler J, Wentworth D, Neaton JD. Is relationship between serum cholesterol and risk of premature death from coronary heart disease continuous and graded? Findings in 356,222 primary screenees of the Multiple Risk Factor Intervention Trial (MRFIT). JAMA. 1986 Nov 28;256(20):2823-8.
- The Lipid Research Clinics Coronary Primary Prevention Trial results. II. The relationship of reduction in incidence of coronary heart disease to cholesterol lowering. JAMA. 1984 Jan 20;251(3):365-74.
- Nissen SE et. al. GAUSS-3 Investigators. Efficacy and Tolerability of Evolocumab vs Ezetimibe in Patients With Muscle-Related Statin Intolerance: The GAUSS-3 Randomized Clinical Trial. JAMA. 2016 Apr 19;315(15):1580-90.
- Vallejo-Vaz AJ, Robertson M, Catapano AL, Watts GF, Kastelein JJ, Packard CJ, Ford I, Ray KK. Low-Density Lipoprotein Cholesterol Lowering for the Primary Prevention of Cardiovascular Disease Among Men With Primary Elevations of Low-Density Lipoprotein Cholesterol Levels of 190 mg/dL or Above: Analyses From the WOSCOPS (West of Scotland Coronary Prevention Study) 5-Year Randomized Trial and 20-Year Observational Follow-Up. Circulation. 2017 Nov 14;136(20):1878-1891.
- Ford I, Murray H, McCowan C, Packard CJ. Long-Term Safety and Efficacy of Lowering Low-Density Lipoprotein Cholesterol With Statin Therapy: 20-Year Follow-Up of West of Scotland Coronary Prevention Study. Circulation. 2016 Mar 15;133(11):1073-80.
- Franklin BA, Brubaker P, Harber MP, Lavie CJ, Myers J, Kaminsky LA. The Journal of Cardiopulmonary Rehabilitation and Prevention at 40 yr and Its Role in Promoting Preventive Cardiology: Part 2. J Cardiopulm Rehabil Prev. 2020 Jul;40(4):209-214.
- Álvarez C, Ramírez-Campillo R, Lucia A, Ramírez-Vélez R, Izquierdo M. Concurrent exercise training on hyperglycemia and comorbidities associated: Non-responders using clinical cutoff points. Scand J Med Sci Sports. 2019 Jul;29(7):952-967.
- Bakker EA, Lee DC, Sui X, Eijsvogels TMH, Ortega FB, Lee IM, Lavie CJ, Blair SN. Association of Resistance Exercise With the Incidence of Hypercholesterolemia in Men. Mayo Clin Proc. 2018 Apr;93(4):419-428.